RESULTS
This comparative study involved 150 participants, consisting of 136 males and 14 females, aged between 18 and 80 years. The subjects were divided into three groups: Group A (control), Group B (precancerous), and Group C (cancerous), with 50 subjects in each group based on specific inclusion and exclusion criteria. The aim was to estimate the levels of SOD and GPx in oral precancerous and cancerous lesions. The age ranges for each group were as follows: Group A ranged from 38 to 69 years, Group B from 28 to 78 years, and Group C from 26 to 72 years. The mean ages (± SE) for each group were 50.72 ± 0.89 years for Group A, 47.12 ± 1.69 years for Group B, and 49.92 ± 1.69 years for Group C, with median ages of 50 years, 45 years, and 50 years, respectively. In terms of the gender distribution, Group A had five females (10.0%) and 45 males (90.0%), Group B comprised seven females (14.0%) and 43 males (86.0%), while Group C included two females (4.0%) and 48 males (96.0%).
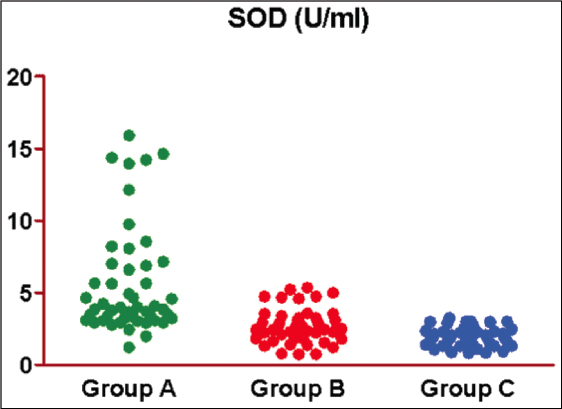
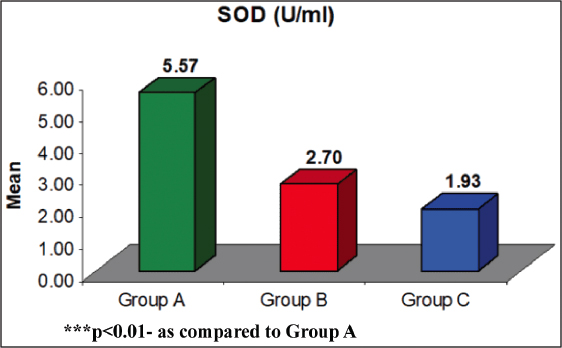
In this study, the SOD levels (measured in U/ml) were observed in three groups: Group A, Group B, and Group C. The SOD levels ranged from 1.23 to 15.91 for Group A, 0.74 to 5.36 for Group B, and 0.83 to 3.27 for Group C. The means (± SE) for each group was as follows: Group A had a mean of 5.57 ± 0.52, Group B had a mean of 2.70 ± 0.16, and Group C had a mean of 1.93 ± 0.09. The medians for the groups were 3.9 for Group A, 2.6 for Group B, and 1.9 for Group C. The results indicate that the mean SOD level was highest in Group A, followed by Group B, and lowest in Group C (Group A > Group B > Group C). This trend suggests that as the severity of the condition increases, the mean SOD levels decrease [Figure 1 and Table 2].
Figure 1: Scatterplot showing the observed SOD of three groups
Table 2: Basic characteristics of three groups
| Basic characteristics | Group A (n = 50) (%) | Group B (n = 50) (%) | Group C (n = 50) (%) | F/χ2 | P value value |
|---|---|---|---|---|---|
| Age (years) | |||||
| Mean ± SE | 50.72 ± 0.89 | 47.12 ± 1.69 | 49.92 ± 1.69 | 1.59 | 0.207 |
| Sex | |||||
| Female | 5 (10.0) | 7 (14.0) | 2 (4.0) | ||
| Male | 45 (90.0) | 43 (86.0) | 48 (96.0) | 2.99 | 0.224 |
The mean differences in SOD between the groups were observed. The Tukey test showed a significantly different and lower SOD in both Group B (51.5%) (5.57 ± 0.52 vs. 2.70 ± 0.16, q = 8.97, P < 0.001) and Group C (65.3%) (5.57 ± 0.52 vs. 1.93 ± 0.09, q = 11.31, P < 0.001) as compared to Group A [Table 3 and Figure 2]. However, it did not differ between Group B and Group C (2.70 ± 0.16 vs. 1.93 ± 0.09, q = 2.34, P = 0.226), although it was lower (28.4%) in Group C as compared to Group B [Table 3 and Figure 3].
Table 3: SOD (U/ml) of three groups
| Group | Min | Max | Mean | SE | Median |
|---|---|---|---|---|---|
| Group A | 1.23 | 15.91 | 5.57 | 0.52 | 3.9 |
| Group B | 0.74 | 5.36 | 2.70 | 0.16 | 2.6 |
| Group C | 0.83 | 3.27 | 1.93 | 0.09 | 1.9 |
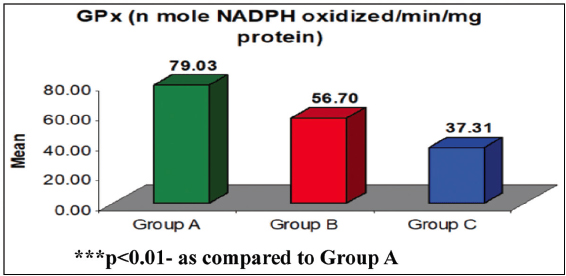
Figure 2: Mean GPx of three groups. ***P < 0.01 – as compared to Group A
Figure 3: Mean SOD of three groups. ***P < 0.01 – as compared to Group A
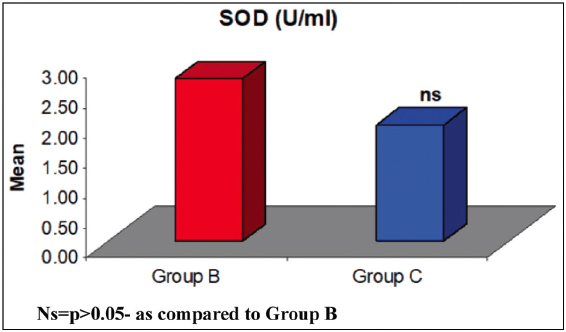
The difference in the mean SOD between groups were compared by Tukey test showing significantly different SOD between Group A and Group B (q = 8.97, P < 0.001) and Group A and Group C (q = 11.31, P < 0.001) but similar between Group B and Group C (q = 2.34, P = 0.226) [Figure 4].
Figure 4: Comparison of mean SOD between two groups. Ns = P > 0.05 – as compared to Group B
The observed GPx (n mole NADPH oxidized/min/mg protein) of three groups is presented in Table 4 and Figure 5.
Table 4: Comparison of mean differences in SOD between the groups by Tukey test
| Comparison | Mean difference | q value | P value | 95% CI (mean difference) |
|---|---|---|---|---|
| Group A vs. Group B | 2.87 | 8.97 | <0.001 | 1.80 to 3.95 |
| Group A vs. Group C | 3.62 | 11.31 | <0.001 | 2.55 to 4.70 |
| Group B vs. Group C | 0.75 | 2.34 | 0.226 | −0.32 to 1.82 |
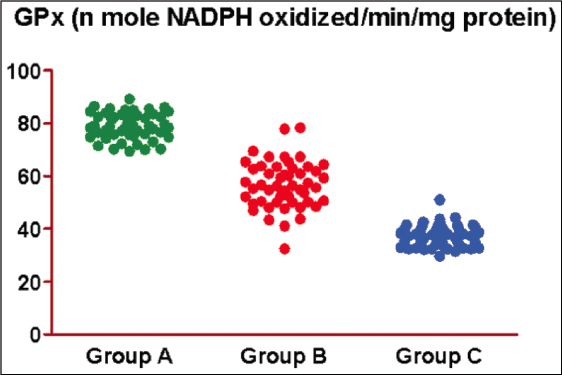
Figure 5: Scatter plot showing observed GPx of three groups
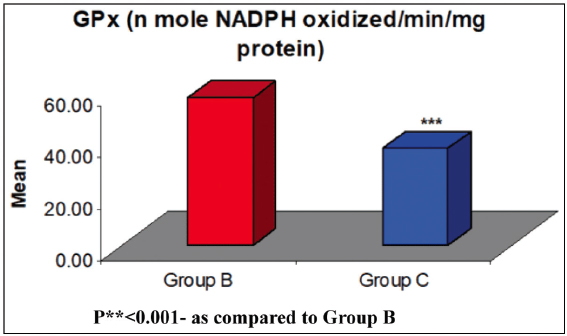
It has been observed that the GPx of Group A, Group B, and Group C ranged from 69.42–89.24, 32.51–78.39, and 29.70–51.08, respectively, with mean (± SE) 79.03 ± 0.70, 56.70 ± 1.26, and 37.31 ± 0.59, respectively, and median 78.8, 56.3, and 37.3, respectively. The mean GPx of Group A was the highest, followed by Group B, and Group C the least (Group A > Group B > Group C) In other words, as severity increases, the mean GPx decreases. It has been found that as regards the mean difference in GPx between the groups, Tukey’s test showed significantly different and lower GPx in both Group B (28.3%) (79.03 ± 0.70 vs. 56.70 ± 1.26, q = 24.90, P < 0.001) and Group C (52.8%) (79.03 ± 0.70 vs. 37.31 ± 0.59, q = 46.51, P < 0.001) as compared to Group A. Furthermore, it was also reduced significantly in Group C (34.2%) as compared to Group B (56.70 ± 1.26 vs. 37.31 ± 0.59, q = 21.61, P < 0.001) [Table 5 and Figure 2].
Table 5: GPx (n mole NADPH oxidized/min/mg protein) of three groups
| Group | Min | Max | Mean | SE | Median |
|---|---|---|---|---|---|
| Group A | 69.42 | 89.24 | 79.03 | 0.70 | 78.8 |
| Group B | 32.51 | 78.39 | 56.70 | 1.26 | 56.3 |
| Group C | 29.70 | 51.08 | 37.31 | 0.59 | 37.3 |
The differences in mean GPx between groups were compared by Tukey’s test and showed significantly different GPx between Group A and Group B (q = 24.90, P < 0.001), Group A and Group C (q = 46.51, P < 0.001), and Group B and Group C (q = 21.61, P < 0.001) [Figures 6 and 7, and Table 6].
Figure 6: Comparison of mean GPx between two groups. P** < 0.001 – as compared to Group B
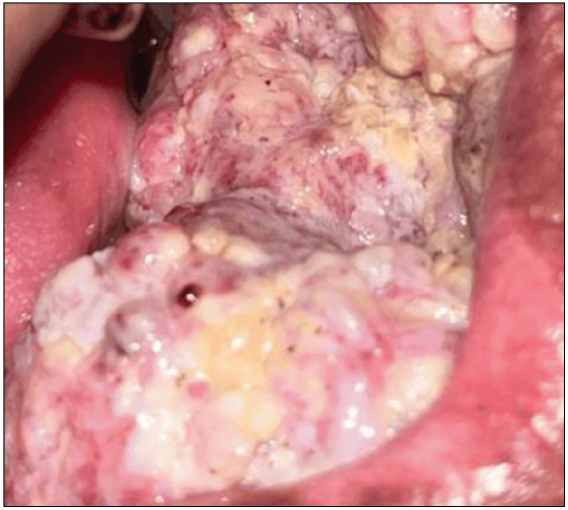
Figure 7: Clinical image of an oral cancer, included in this study
Table 6: Comparison of mean difference in GPx between the groups by Tukey’s test
| Comparison | Mean difference | q value | P value | 95% CI (mean difference) |
|---|---|---|---|---|
| Group A vs. Group B | 22.33 | 24.90 | <0.001 | 19.32 to 25.34 |
| Group A vs. Group C | 41.71 | 46.51 | <0.001 | 38.70 to 44.72 |
| Group B vs. Group C | 19.38 | 21.61 | <0.001 | 16.37 to 22.39 |